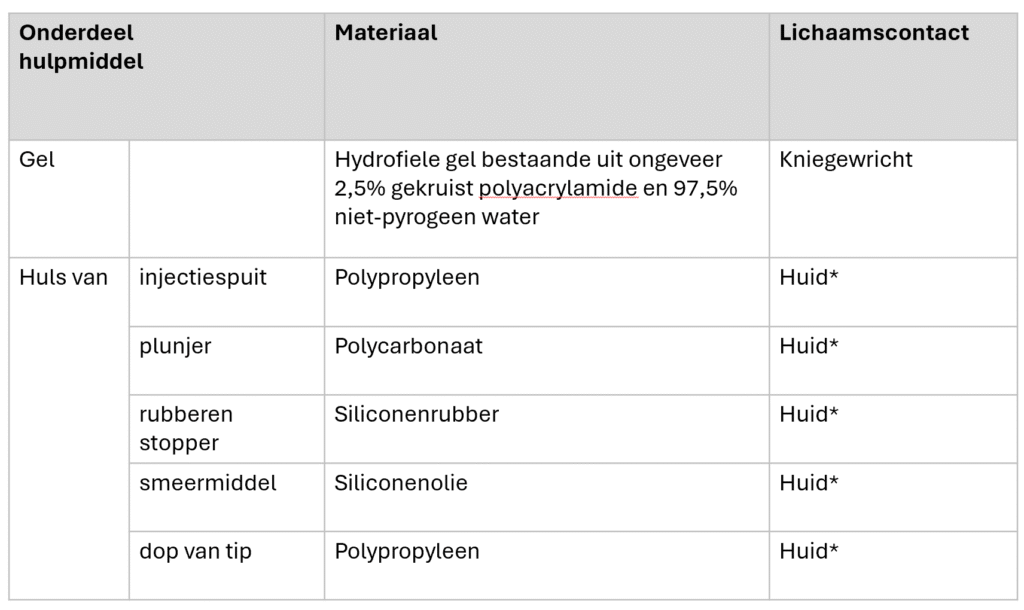
As described above, Arthrosamid®hydrogel consists of 2.5% dry matter crosslinked polyacrylamide and 97.5% non-pyrogenic water. During synthesis, N,N, methylene-bis-acrylamide polymerizes with acrylamide, forming cross-links between polyacrylamide chains.
The crosslinked polyacrylamide hydrogel is synthesized in a polymerization process in which repeating units of acrylamide are linked together in a chain reaction.
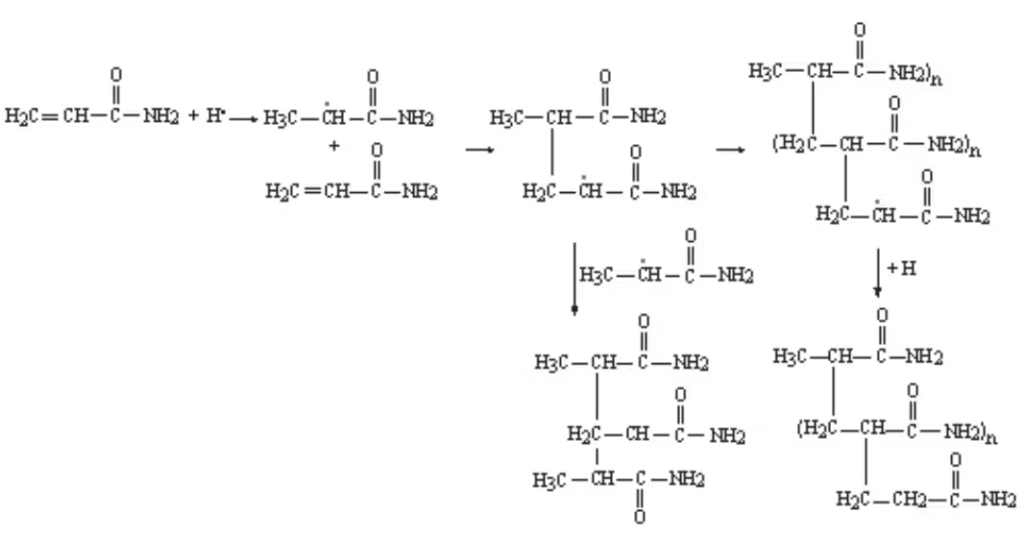
The chain reaction uses a redox initiator principle, in which an ammonium persulfate initiator (AMPS) generates a free radical that adds an acrylamide monomer by obtaining an electron from the double bond in the acrylamide, forming a single carbon-carbon bond with the acrylamide. This leaves an undivided electron for further addition of acrylamide monomers, as shown in Figure 2 on the right.
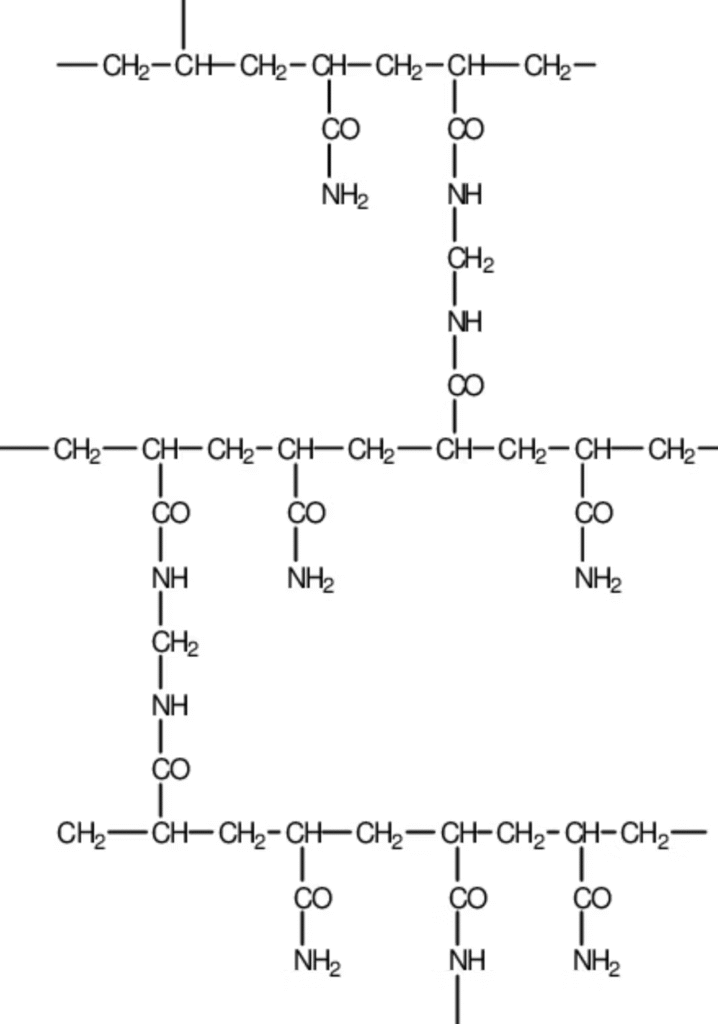
Branching of the linear carbon-carbon structure of polyacrylamide will occur due to the presence of a crosslinked agent, N,N-methylene-bis-acrylamide monomer (MBAM), as shown in Figure 3.
After branching of the polyacrylamide chains of the structure, a three-dimensional network of crossed polyacrylamide is formed, rather than non-coupled linear chains of polyacrylamide.
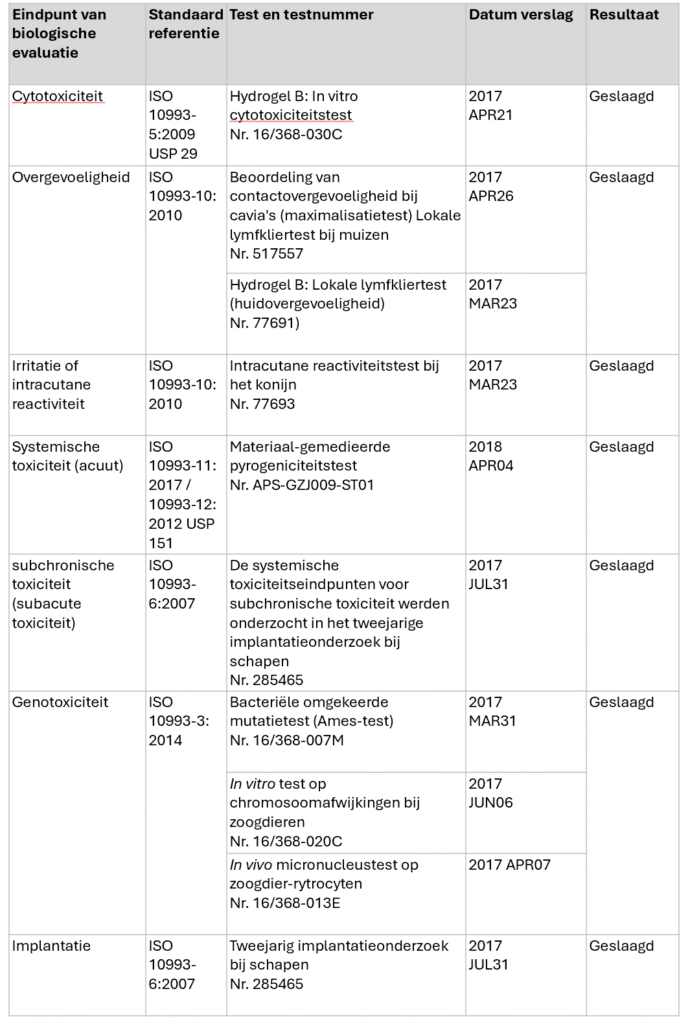
Arthrosamid® is supplied as a pre-filled, sterile 1 mL single-use syringe sealed with a luer lock fitting and a sealing cap. The drug is intended to be injected intra-articularly into the knee joint with a 21G x 2 inch (0.8 x 50 mm) sterile needle.
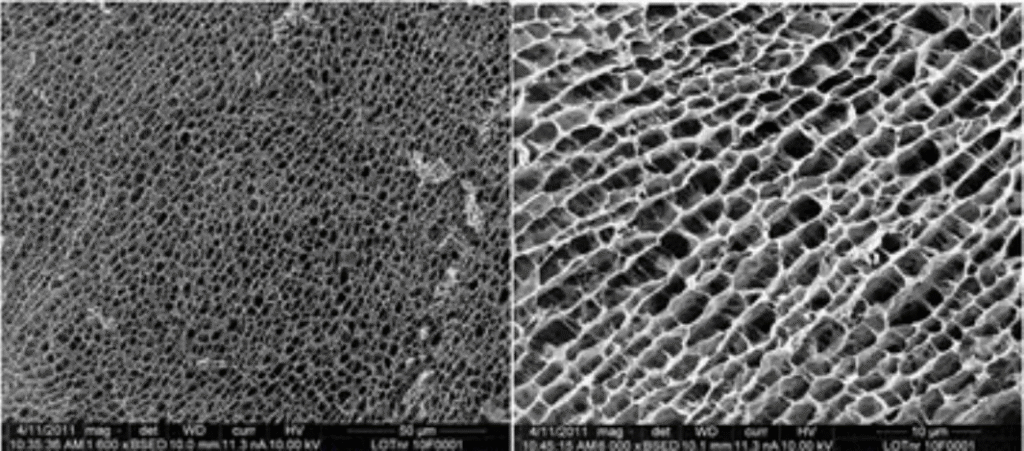
Figure 1 shows the three-dimensional structure of the polyacrylamide hydrogel. The tight uniform honeycomb structure provides a grid for cell growth.