Arthrosamid® is an injectable hydrogel consisting of 97.5% water and 2.5% crosslinked polyacrylamide. Treatment is via six separate 1 ml injections, where the hydrogel is introduced into the synovial space. This leads to the following physiological changes:
✔ Tissue growth and thickening – After injection, synovial tissue grows in the open structure of Arthrosamid®, thickening and strengthening the joint capsule.
✔ Permanent presence – Arthrosamid® is not resorbed or degraded, but remains permanently present in the tissue.
✔ S afety and long-term use – More than 1,800,000 treatments have been performed worldwide with no permanent adverse side effects.
✔ Knee replacement possible after treatment – In a 10-year retrospective study of 120 patients, 56% did not need a knee replacement after 10 years. For the remaining patients who received a prosthesis, there were no side effects related to the hydrogel.
✔ Improved tissue elasticity – Clinical studies show a 15% increase in synovial tissue elasticity, normalizing joint function.
✔ Reduced pain signals – Improved elasticity reduces nocisensory pain perception, leading to reduced pain symptoms.
✔ Anti-inflammatory effect – Arthrosamid® thickens synovial tissue and creates a natural barrier to inflammatory cells, reducing inflammatory activity, improving mobility and reducing pain symptoms.
✔ Healthier synovial fluid – Recent research (2025) shows that patients with a positive response have more hyaluronic acid and less interleukin-6 (IL-6) in their synovial fluid, indicating reduced pro-inflammatory activity.
Pain reduction
Mobility
Functionality
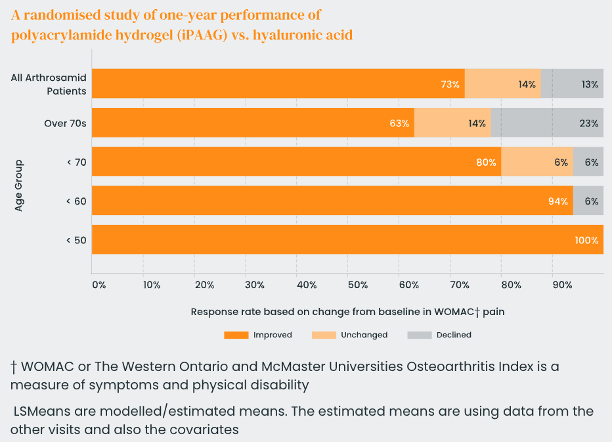
Arthrosamid® intra-articular (IA) polyacrylamide gel (iPAAG) is safe for its intended use in the treatment of pain symptoms in knee osteoarthritis. The most common adverse events (side effects) in clinical trials were injection-related mild to moderate pain and or mild swelling during the first weeks after injection.
More than 1,800,000 treatments have now been performed with this hydrogel over the past 25 years with no known negative side effects that can be related to the hydrogel.
An observational long-term study examined the safety of intra-articular 2.5% polyacrylamide hydrogel (iPAAG) in patients with knee osteoarthritis (OA). The study followed 61 patients (89 knees) for 7 to 14 years (mean 9.92 years). The mean age at injection was 64 years.
No significant adverse events were reported related to iPAAG, including no allergies, infections or systemic effects.
43.8% of treated knees eventually underwent knee replacement, on average after 3.4 years.
56.2% of knees did not need a prosthesis even after 10 years, after only one treatment.
Surgical complications in subsequent knee replacements were minimal and not related to iPAAG.
Results confirm a favorable long-term safety profile. iPAAG appears to be a reliable, non-surgical treatment option for knee osteoarthritis, with potential to delay knee replacements for a long time.
For further results, please refer to the clinical data where all literature is listed with a link/reference to the article.
Op arthrosamid.be gebruiken wij cookies om de website goed te laten functioneren en om uw gebruikerservaring te verbeteren. U kunt kiezen welke cookies u wilt toestaan. Bekijk voor meer informatie ons privacybeleid.